ABX has received marketing authorisation for the radiotracer Radelumin® in Germany
ABX has received marketing authorisation for the radiotracer Radelumin® in Germany! This marks the 8th European country including the Netherlands in which the authorisation has been obtained. In France, Radelumin® is already in application since December 2022 – production and marketing in the Netherlands will start by the beginning of 2024. Radelumin will be manufactured at RTM for the Dutch market.
Radelumin® successfully passed a clinical phase III study (ABX-CT-301). It was subsequently approved in France at the end of 2021. ABX then started a Mutual Recognition Procedure (MRP), in order to obtain the marketing authorisation in further European countries. The MRP process was decided positively in March 2023 and since then, seven European countries – Austria, Belgium, Italy, Luxembourg, Spain, The Netherlands and now Germany – approved Radelumin®.
Radelumin® ([18F]PSMA-1007) was a follow-up to the success of [177Lu]PSMA-617 in 2018 by the Deutsche Krebsforschungszentrum (DKFZ). It was developed with the support of Prof. Dr. Frederik L. Giesel, director of the Clinic for Nuclear Medicine at the University Hospital Düsseldorf, as well as Prof. Dr. Klaus Kopka, director of the Institute of Radiopharmaceutical Cancer Research at the Helmholtz Center Dresden-Rossendorf and formerly head of Radiopharmaceutical Chemistry at the DKFZ.
The compound is injected into the patient and binds to the prostate-specific membrane antigen (PSMA) – a cell surface protein which is highly overexpressed in prostate cancer cells. The radioactive fluorine-18 subsequently makes the cancer visible for positron emission tomography (PET) and at the same time decays quickly in the body. Due to its radioactive short-lived nature, the production and application of Radelumin® needs extensive preparation of supply chains. Production sites, hospitals and partners from all over the European continent are in the process of being onboarded; marketing and application of Radelumin® on a grand scale is expected to launch beginning of 2024.
More information: abx.de
Other news

Huntington’s Disease: A Review of the Known PET Imaging Biomarkers and Targeting Radiotracers
Klaudia Cybulska (postdoc radiochemistry) has written an article about Huntington's Disease. Abstra...
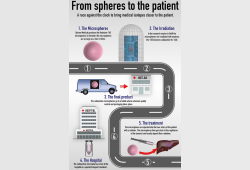
First liver cancer patient treated with microspheres irradiated in TU Delft
Radboud Translational Medicine will perform the dispensing of below product. The production and dis...

RTM receives marketing authorisation for Fludeoxyglucose (18F)
We are happy to announce that RTM has received marketing authorization for 18F-FDG of 17 July 2018....
First Patient Dosed in Phase II Study of TLX250-CDx in Triple-Negative Breast Cancer
The below product is manufactured at RTM First Patient Dosed in Phase II Study of TLX250-CDx in Tri...
Interested in a collaboration?
Contact us to discuss the possibilities.





