RTM supports Citryll’s imaging trial of [89Zr]Zr-DFO-CIT-013
Radboud Translational Medicine (RTM) is proud to announce its role as manufacturing partner for the clinical imaging trial of [89Zr]Zr-DFO-CIT-013, a radiolabeled version of CIT-013 developed by Citryll.
Citryll is a biotech company pioneering a transformative approach to treating immune-mediated inflammatory diseases by targeting Extracellular Traps (ETs), a key driver of inflammation. The imaging trial, ‘Cityview’, is designed to assess the biodistribution of radiolabeled CIT-013 using PET-CT imaging in participants with rheumatoid arthritis (RA) and hidradenitis suppurativa (HS). The trial will provide important insights into target engagement and the mechanism of action of this first-in-class antibody targeting ETs. The clinical team conducting the trial operates within the Radboudumc Departments of Rheumatology and Medical Imaging, with close involvement from Dermatology, forming a multidisciplinary collaboration focused on patient recruitment and the efficient execution of the clinical trial.
RTM has established a robust and scalable GMP-compliant manufacturing process for [89Zr]Zr-DFO-CIT-013 and successfully completed process validation. In addition, the company provided scientific expertise in radiopharmaceutical development and GMP manufacturing, as well as the required CMC data package to support clinical trial approval.
Following these achievements, RTM is responsible for the routine GMP manufacturing and supply of [89Zr]Zr-DFO-CIT-013 for the ongoing clinical trial.
This collaboration highlights RTM’s capabilities in the development, validation, and clinical supply of innovative radiopharmaceuticals, supporting biotech partners from early development through clinical application.
For more information on Citryll’s imaging trial, read the full announcement: First patient dosed in Citryll’s imaging study evaluating distribution and target engagement of CIT-013 - Citryll
![RTM supports Citryll’s imaging trial of [89Zr]Zr-DFO-CIT-013](https://radboudtranslationalmedicine.nl/cdn/42251bd5-d9e6-4107-bbd7-ac13f0c8609a/-/resize/555x/-/format/auto/-/quality/smart/citryll-cityview-combined-logo.png)
Other news

LinkedIn post Isologic goes electric
🔌Isologic Goes Electric! We are excited to announce the expansion of our electric fleet with the a...

LinkedIn post ESRR2024
📢 Important update on EU #GMP Annex 1 at #ESRR2024! The revised EU GMP Annex 1, effecti...
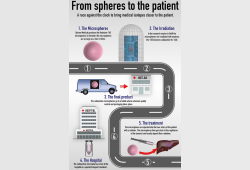
First liver cancer patient treated with microspheres irradiated in TU Delft
Radboud Translational Medicine will perform the dispensing of below product. The production and dis...

18F-FDOPA now available
Fluorodopa (18F) is now available for clinical studies and - with doctor's certificate - for routine...
Interested in a collaboration?
Contact us to discuss the possibilities.





