RTM receives marketing authorisation for Fludeoxyglucose (18F)
We are happy to announce that RTM has received marketing authorization for 18F-FDG of 17 July 2018.
Fludeoxyglucose (18F) RTM 200 MBq/ml, solution for injection will become commercially available in The Netherlands in October, with roll-out to Belgium and Germany planned in 2019.

Other news
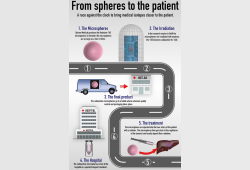
First liver cancer patient treated with microspheres irradiated in TU Delft
Radboud Translational Medicine will perform the dispensing of below product. The production and dis...

GMP-certificate received
Today we received our GMP-certificate!The results of the inspection were excellent and no shortcomin...
First Patient Dosed in Phase II Study of TLX250-CDx in Triple-Negative Breast Cancer
The below product is manufactured at RTM First Patient Dosed in Phase II Study of TLX250-CDx in Tri...

Production of novel diagnostic radionuclides in small medical cyclotrons
Employees from RTM and Radboudumc have published a new article: 'Production of novel diagnostic radi...
Interested in a collaboration?
Contact us to discuss the possibilities.





