First Patient Dosed with TLX250-CDx in Italy for ccRCC
The below product is manufactured at RTM
First Patient dosed with TLX250-CDx in Italy for ccRCC
The article below was published on March 25, 2024 at www.urologytimes.com by Hannah Clarke:
With the dosing of the first patient in Italy, Telix’s expanded access program for TLX250-CDx in ccRCC now includes 3 active countries, including the Netherlands and the US.
The first patient in Italy has been dosed with TLX250-CDx (89Zr-DFO-girentuximab, Zircaix) through the global expanded access (“accesso allargato”) program for patients with clear cell renal cell carcinoma (ccRCC), announced Telix Pharmaceuticals, the developer of the PET imaging agent, in a news release.1
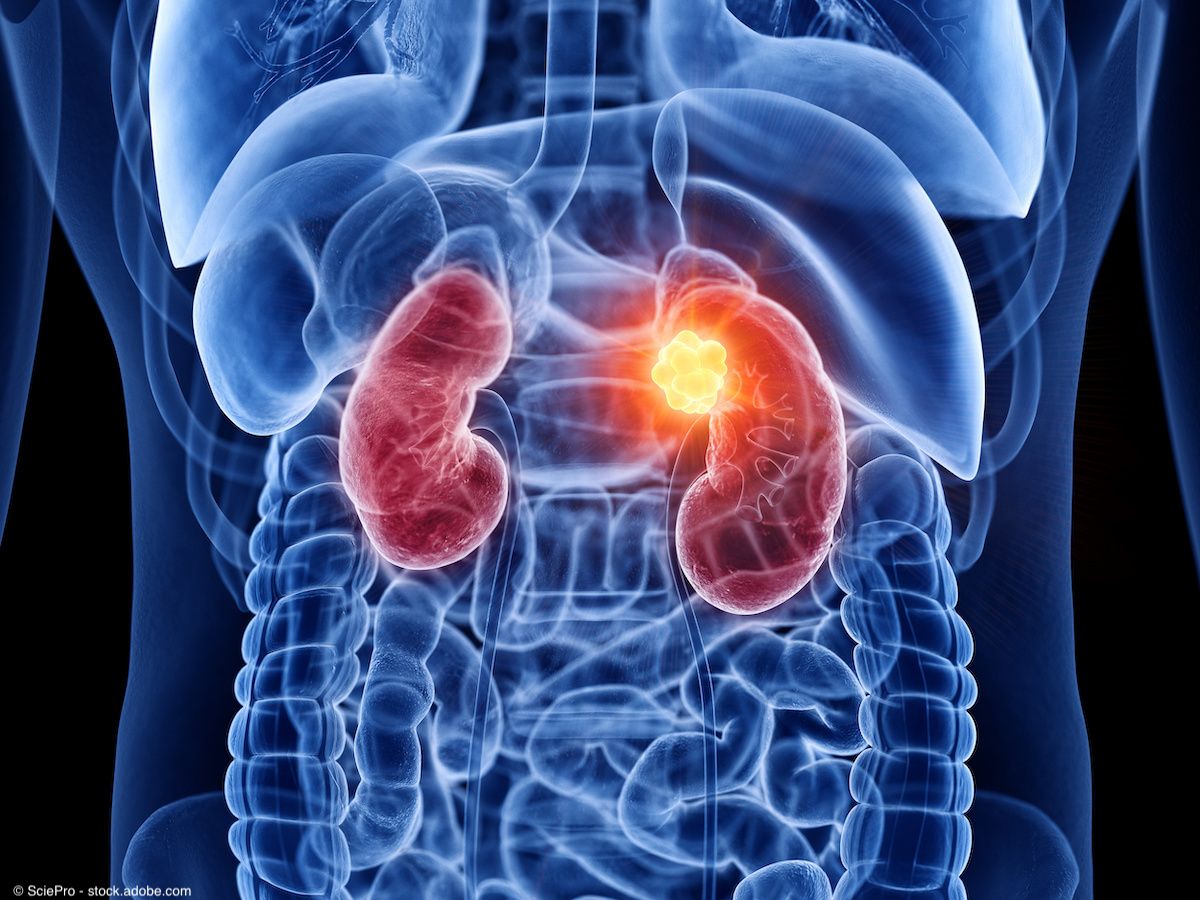
The performance of TLX25-CDx in ccRCC was explored in the phase 3 ZIRCON trial, which reported positive results in November 2022.
The first patient was dosed with TLX250-CDx at RCCS Ospedale San Raffaele in Milan, Italy.
"Continued access in Europe to TLX250-CDx is vitally important to enable early detection of ccRCC, which can often be challenging and reliant on invasive biopsy and nephrectomy (kidney removal). In TLX250-CDx, I am extremely pleased to be able to offer a non-invasive 'Molecular Biopsy' to address unmet need in my patients in Italy," said Arturo Chiti in the news release.1 Chiti is a professor of diagnostic imaging and radiotherapy at IRCCS Ospedale San Raffaele.
With the dosing of the first patient in Italy, Telix’s expanded access program now includes 3 active countries, including the Netherlands and the US, both of which first dosed patients in December 2023. The program allows access to TLX250-CDx for patients outside of a clinical trial for whom there is no comparable or satisfactory alternative options.
Telix also submitted a biologics license application to the US FDA for TLX25-CDx in ccRCC in December 2023, with hopes of commercial availability in 2024. The company is progressing with equivalent applications with other regulatory agencies as well.
"We are pleased to support unmet patient need by adding a second European country to Telix's global expanded access program for TLX250-CDx. Through the ZIRCON study, this investigational agent has demonstrated high sensitivity and specificity in the detection of ccRCC with potential to change standard of care in the diagnosis and management of this most aggressive form of kidney cancer,” said Frédéric Fantino, Telix medical director for the Europe, Middle East and Africa region, in the news release.1
Data on TLX250-CDx
The performance of TLX25-CDx in ccRCC was explored in the phase 3 ZIRCON trial (NCT03849118), which reported positive results in November 2022.2
Overall, the trial met its primary and secondary end points by demonstrating high specificity and sensitivity of the agent in adult patients with ccRCC and an indeterminate renal mass (IDRM). In the detection of the clear cell phenotype and diagnosing the presence and spread of ccRCC, TLX250-CDx achieved a sensitivity of 86% and a specificity of 87%, meeting the trial’s co-primary end points. The secondary end point was also met with an 85% sensitivity and 90% specificity of the agent in detecting ccRCC tumors less than 4 cm.
Among all evaluable patients, the positive predictive value was 91.7% or higher, and the negative predictive value was 73.7% or higher.3
In total, the open-label, multicenter trial included 300 patients (mean age = 62) who were dosed with TLX250-CDx. Of those, 284 evaluable patients were included in the primary analysis.
To be eligible for the ZIRCON study, patients had to have a single IDRM of 7 cm or smaller in diameter (stage cT1) as identified on CT or MRI that was suspicious for ccRCC and scheduled for surgical removal within 90 days.
For the trial, each patient received a single dose of TLX250-CDx (37 MBq ± 10%; 10 mg girentuximab) via intravenous administration on day 0 and PET/CT imaging on day 5 (±2 days), prior to partial nephrectomy. PET/CT images were assessed by 3 blinded independent readers.
References
1. First patient dosed in Italian named patient (Early Access) Program for TLX250-CDx, Telix's kidney cancer imaging agent. News release. Telix Pharmaceuticals Limited. Published online and accessed March 25, 2024. https://www.prnewswire.com/new...
2. Telix announces positive topline results of ZIRCON phase III kidney cancer imaging study. News release. November 6, 2022. Accessed March 25, 2024. https://www.prnewswire.com/new...
3. Shuch BM, Pantuck AJ, Bernhard JC, et al. Results from phase 3 study of 89Zr-DFO-girentuximab for PET/CT imaging of clear cell renal cell carcinoma (ZIRCON). J Clin Oncol. 2023. doi:10.1200/JCO.2023.41.6_suppl.LBA60
Other news
First Patient Dosed in Phase II Study of TLX250-CDx in Triple-Negative Breast Cancer
The below product is manufactured at RTM First Patient Dosed in Phase II Study of TLX250-CDx in Tri...
ABX has received marketing authorisation for the radiotracer Radelumin® in Germany
ABX has received marketing authorisation for the radiotracer Radelumin® in Germany! This marks the 8...

Huntington’s Disease: A Review of the Known PET Imaging Biomarkers and Targeting Radiotracers
Klaudia Cybulska (postdoc radiochemistry) has written an article about Huntington's Disease. Abstra...

1st production QuiremSpheres
On May 21st, the first patient dosis of QuiremSpheres was processed at RTM and then shipped to the s...
Interested in a collaboration?
Contact us to discuss the possibilities.





